Toronto, Canada: June 19–20, 2025 — Altis Labs, Inc. ("Altis") is announcing results presented at the Canadian Breast Cancer Symposium (CBCS) 2025 evaluating the prognostic utility of Imaging-based Prognostication (IPRO), an AI-derived score from pretreatment CT imaging, in patients with breast cancer.
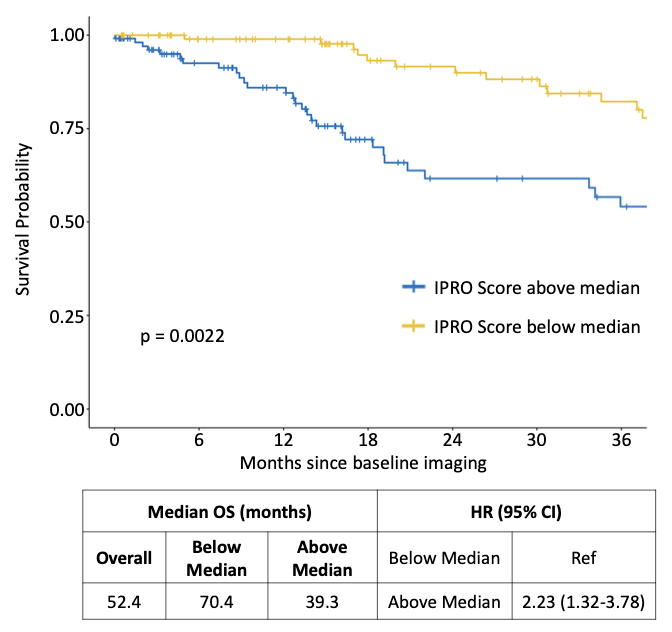
The study retrospectively evaluated IPRO against TNM stage and patient age in a cohort of 219 breast cancer patients with a pre-treatment CT scan available, diagnosed at a single academic cancer center in Ontario from 2014–2022. The cohort included 172 early-stage (I–IIIA) and 47 advanced-stage (IIIB–IV) patients, with a median patient age of 60.8 years and a median OS for the entire population of 52.4 months. Full receptor status was available for 200 patients: 54.1% hormone receptor-positive, HER-2 negative; 14.5% triple-negative; 11.8% triple-positive; and 10.5% hormone receptor-negative, HER-2 positive. Kaplan–Meier methods, hazard ratios, and concordance index (c-index) were used to estimate covariate effects on OS and prognostic accuracy for IPRO (above vs. below median), clinical TNM stage (early vs. advanced), and patient age (above vs. below median).
Key findings:
Notably, IPRO was trained exclusively on advanced non-small cell lung cancer (aNSCLC) thoracic CT scan data — making its performance in breast cancer an out-of-distribution generalization. These results indicate that the imaging biomarkers underpinning IPRO are generalizable across differing tumour types, and provide rationale for further studies in breast cancer. Future work will assess the performance of a version of IPRO trained on breast cancer data, in larger sample sizes, across different treatment modalities, and compared across receptor and molecular subtypes.
About Altis Labs
Altis Labs is the computational imaging company accelerating clinical trials with AI. Altis has trained proprietary AI models on the industry's largest multimodal training database spanning over 200 million longitudinal images linked to clinical, molecular, treatment, and outcomes data. Top 20 biopharmas use Altis' AI models to more confidently analyze data from phase 1–4 clinical trials and bring the most effective novel treatments to patients sooner. For more information, visit www.altislabs.com, follow @AltisLabs on social media, or email info@altislabs.com.